2024年4月29日,美国食品和药物管理局(FDA)授予tisotumab vedotin-tftv(Tivdak)的传统批准,用于治疗化疗期间或化疗后疾病进展的复发性或转移性宫颈癌。
该批准基于全球随机3期innovaTV 301临床试验的结果(NCT04697628),2023年10月,innovaTV 301研究的最初结果在欧洲肿瘤内科学会(ESMO)大会的主席研讨会上披露。
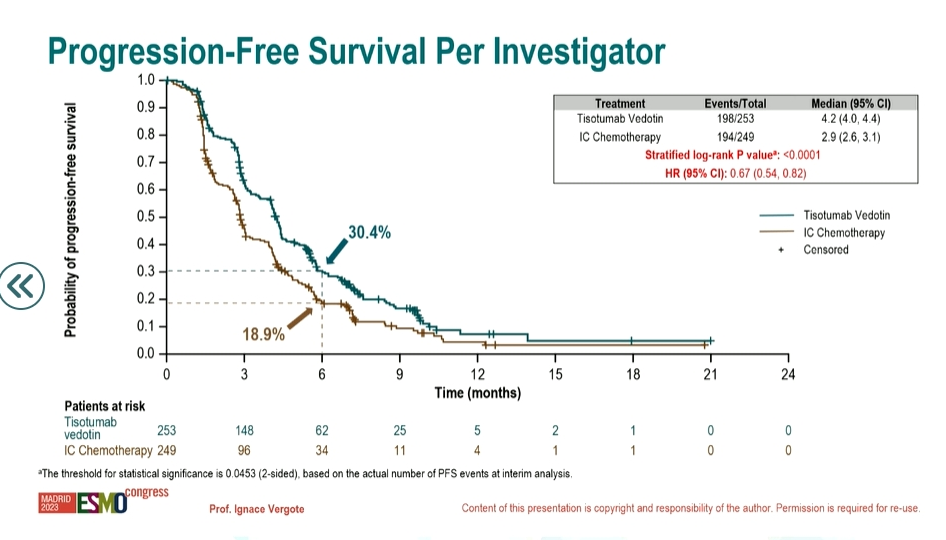
该试验纳入502名既往接受过一种或两种全身治疗方案的复发性或转移性宫颈癌(r/mCC)患者:tisotumab vedotin 组的中位总生存期(OS)为11.5个月,化疗组的中位OS为9.5个月;tisotumab vedotin组的中位无进展生存期(PFS)为4.2个月,化疗组的中位PFS为2.9个月;各组的确认缓解率(ORR)分别为17.8%和5.2%;与化疗相比,tisotumab vedotin 组的死亡风险降低了30%。
宫颈癌是常见的妇科恶性肿瘤之一。据2023年ICO/IARC中国HPV及其相关疾病报告显示,2020年在中国15-44岁女性中,宫颈癌发病率和死亡率均居女性肿瘤第三位,新发病例数约将近11万,死亡病例将近6万,严重威胁女性健康。
TIVDAK(tisotumab vedotin-tftv)是一种抗体-药物偶联物(ADC),由Genmab针对组织因子(TF)的人源单克隆抗体和辉瑞的ADC技术组成,该技术利用蛋白酶可裂解接头,将微管破坏剂单甲基auristatin E(MMAE)共价连接到抗体上。非临床数据表明,tisotumab vedotin-tftv的抗癌活性是由于ADC与表达TF的癌细胞结合,随后ADC-TF复合物内化并通过蛋白水解裂解释放 MMAE。MMAE破坏活跃分裂细胞的微管网络,导致细胞周期停滞和凋亡细胞死亡。在体外,tisotumab vedotin-tftv 还介导抗体依赖性细胞吞噬作用和抗体依赖性细胞毒性。
2021年9月20日,由Seagen和Genmab公司联合开发的ADC-Tivdak获FDA批准上市,用于治疗化疗期间或化疗结束后疾病进展的复发或转移性宫颈癌,Tivdak是美国食品药品监督管理局(FDA)批准的首款治疗宫颈癌的抗体偶联药物,也是全球首个获批靶向组织因子的抗体偶联药物,为复发或转移性宫颈癌患者提供了新的治疗选择。
好消息是,tisotumab vedotin目前在国内多中心开展临床试验,目前正在招募复发性或转移性宫颈癌患者,想申请的病友可以联系康和源免疫之家医学部(400-880-3716)了解详细的出入排标准,并进行初步评估,看是否有机会申请临床试验。
项目名称:在复发性或转移性宫颈癌患者中比较Tisotumab vedotin 与化疗的III期试验
部分入选标准:
1.同意时年龄 ≥18岁,或根据当地法规认为是成人;
2.随机分组前 ECOG 体能状态为0或1;
3.预期寿命至少为3个月。
想要寻求临床试验帮助的患者可提交病理报告、治疗经历等资料至康和源免疫之家医学部(400-880-3716)进行初步评估。
综上所述,此次批准为长期面临有限选择的患者群体开辟了新的治疗途径。
扫描下方二维码,或者直接电话咨询康和源免疫之家医学部(400-880-3716),为癌友患者们分享更多信息。

免责声明:康和源免疫之家为免疫科普平台,文本参考来源于网络,版权归原作者所有。
该文章仅供分享,如涉嫌侵犯您的著作权请联系我们删除,谢谢!
参考资料
1. FDA approves tisotumab vedotin-tftv for recurrent or metastatic cervical cancer | FDA
2. TIVDAK® Supplemental Biologics License Application Accepted for Priority Review by FDA for Patients with Recurrent or Metastatic Cervical Cancer | Pfizer
3. TIVDAK® (tisotumab vedotin-tftv) Receives U.S. FDA Approval to Treat Recurrent or Metastatic Cervical Cancer - Genmab A/S
热门推荐
参加(正高、副高、中级、规培结业、医师定期考核、三基等)考试,难度越来越大,通过率越来越低。今天必须跟大家推荐一款刷题神器:助考之星

题库科目全、题库质量高、超多技能辅助、还可以随时随地学习
资料这么全、操作这么方便,你还等什么?抓紧时间刷题看视频!更多内容咨询客服电话400-880-3716

70-80%的化疗患者出现恶心、呕吐,恶心呕吐是化疗达到最佳治 疗的巨大障碍,10-20%的患者由于恶心呕吐而拒绝化疗。为了减轻患者的痛苦,持续有效完成整个化学治疗,引进了电子止吐仪。可有效地治疗和缓解恶心及呕吐:妊娠呕吐、化疗呕吐、术后呕吐、晕动呕吐,如晕船、晕车、晕机,VR眩晕,高原反应引起的恶心和呕吐。更多内容咨询客服电话400-880-3716

相关推荐
随便看看
- 客观缓解率达71%!BCMA/CD3双抗linvoseltamab获优先审评资格,为多发性骨髓瘤患者提供更多选择
- IN10018片拟纳入突破性治疗
- 重磅获批!靶向TROP2的ADC药物德达博妥单抗(Dato-DXd)上市,HR+/HER2-晚期乳腺癌患者治疗新方案来了
- PRMT5抑制剂CTS3497胶囊获得临床试验许可
- 美国FDA批准retifanlimab联合方案用于肛管鳞状细胞癌
- 百济神州泽布替尼关键性全球3期SEQUOIA试验C组及D组队列新数据亮相2025 ASCO
- 宜联生物与安进达成合作协议,评估ADC药物YL201联合IMDELLTRA™用于广泛期小细胞肺癌的潜力
- 晚期宫颈癌、胃癌患者迎来新选择!肿瘤药物卡度尼利单抗亮相《柳叶刀》
- 卡度尼利联合普络西用于治疗胃癌的Ⅲ期临床完成首例患者入组
- 贝莫苏拜单抗联合安罗替尼II期研究亮相2024 IGCS,子宫内膜癌患者福音











