
关于Breyanzi 典型案例 如何参加CAR-T细胞临床试验 结语
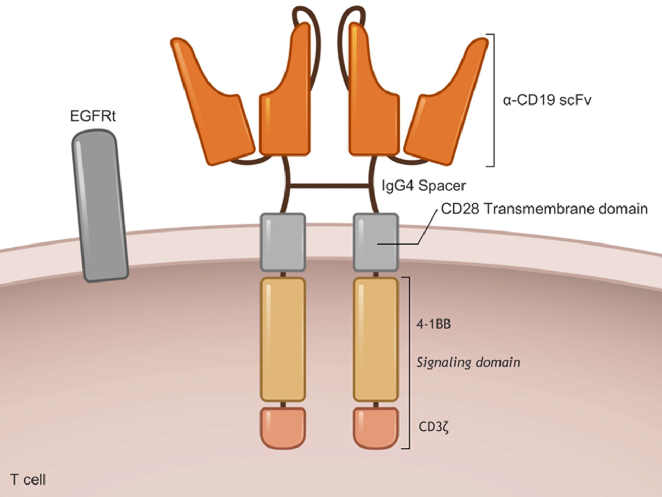


相关推荐
随便看看
- 完全缓解率100%!CAR-T疗法瞄准白血病、淋巴瘤、多发性骨髓瘤
- CAR-T细胞治疗产品纳基奥仑赛新适应症获受理,用于B 细胞淋巴瘤
- 北恒生物靶向CD19的通用型CAR-T细胞产品RD06-03亮相2025 ASCO
- 世界首个!邦耀生物治疗红斑狼疮非病毒定点整合PD1-CAR-T疗法获批中国IND
- CAR-T疗法2.0版来了!“体内CAR-T”疗法,告别百万天价与漫长等待!体内直接编程,癌细胞6小时清零?
- 中位生存近3年!“百万”CAR-T带领淋巴瘤患者走向“治愈”之路
- CAR-T细胞产品纳基奥仑赛实现系统性红斑狼疮相关的免疫性血小板减少症患者实现临床完全缓解
- 邦耀生物CAR-T细胞TyU19成功治疗复发/难治性系统性红斑狼疮
- CAR-T细胞疗法重击结直肠癌、甲状腺癌、卵巢癌等实体瘤
- 全球首个实体瘤CAR-T疗法获批在即!CAR-T让胃癌病灶清零!实体瘤患者迎来曙光











