
12月6日,国家药品监督管理局药品审评中心显示:精准生物自主研发的靶向CD19的CAR-T细胞产品MC-1-50细胞制剂获得临床试验默示许可,用于治疗18岁及以上的难治性系统性红斑狼疮患者。
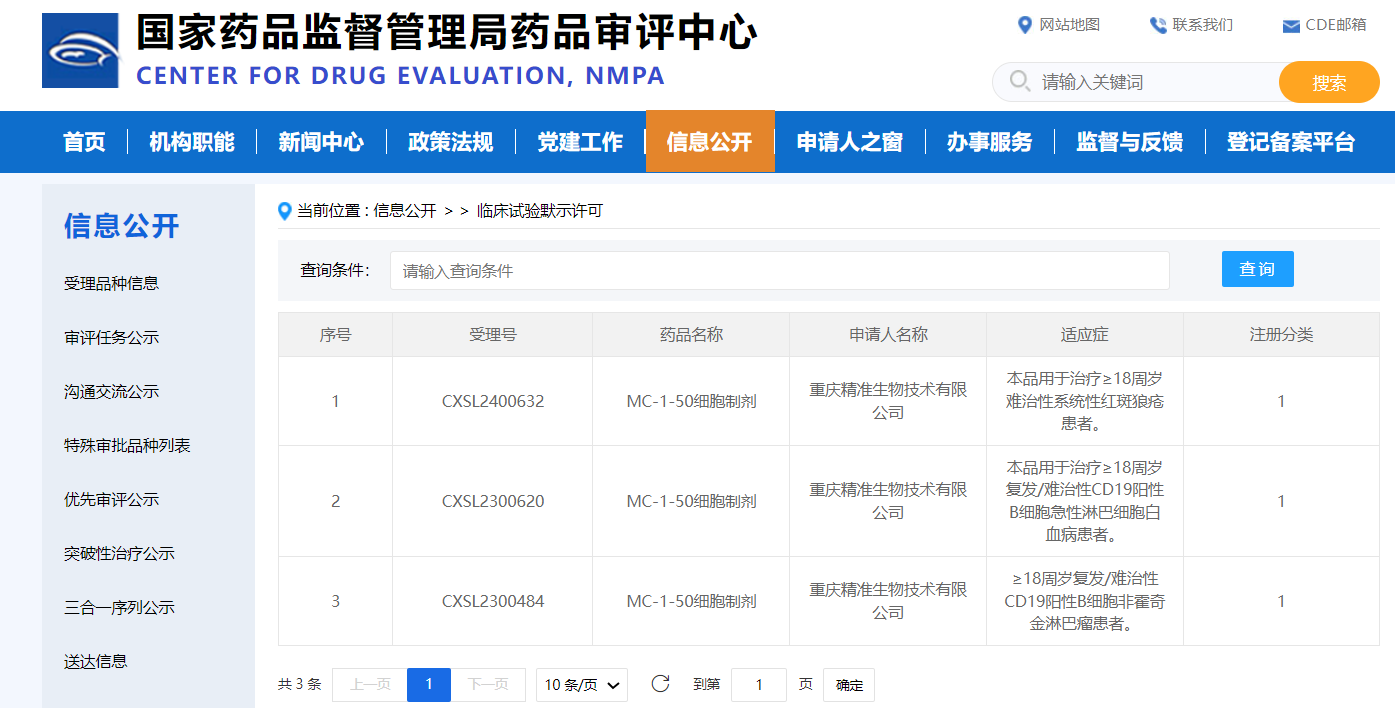

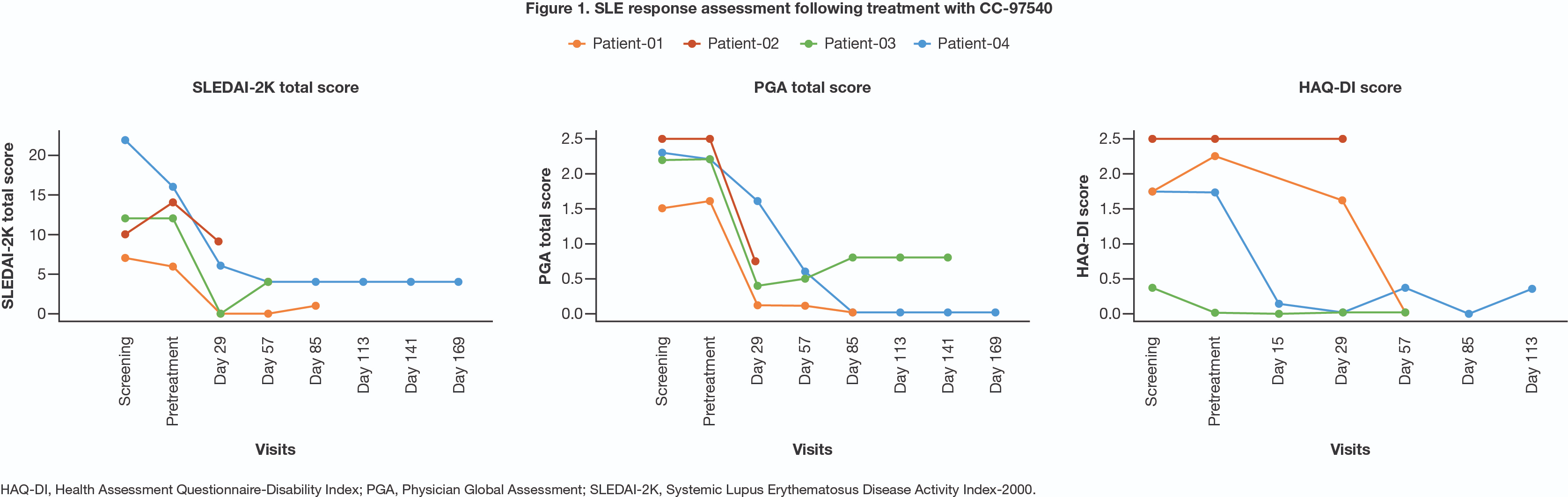
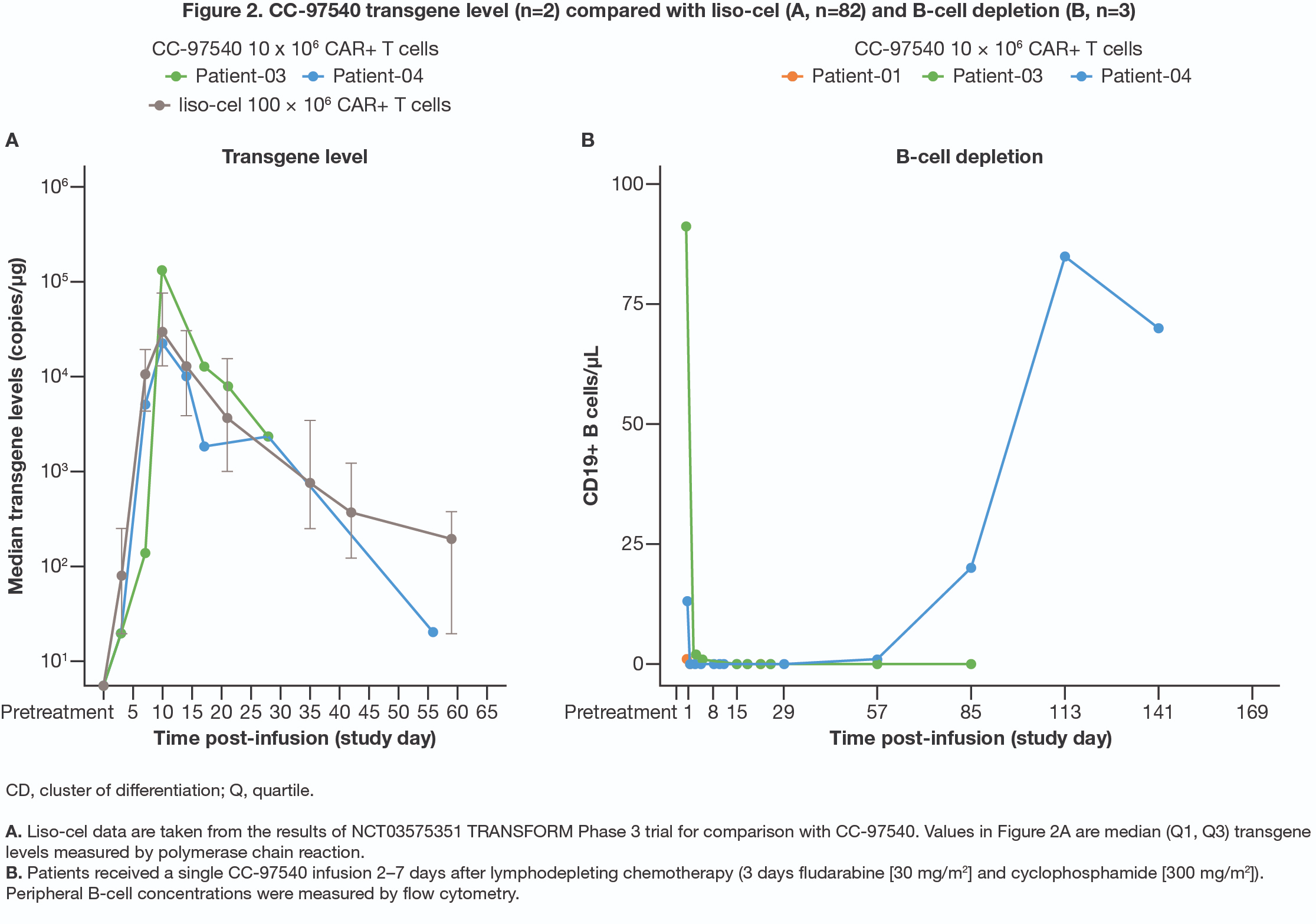
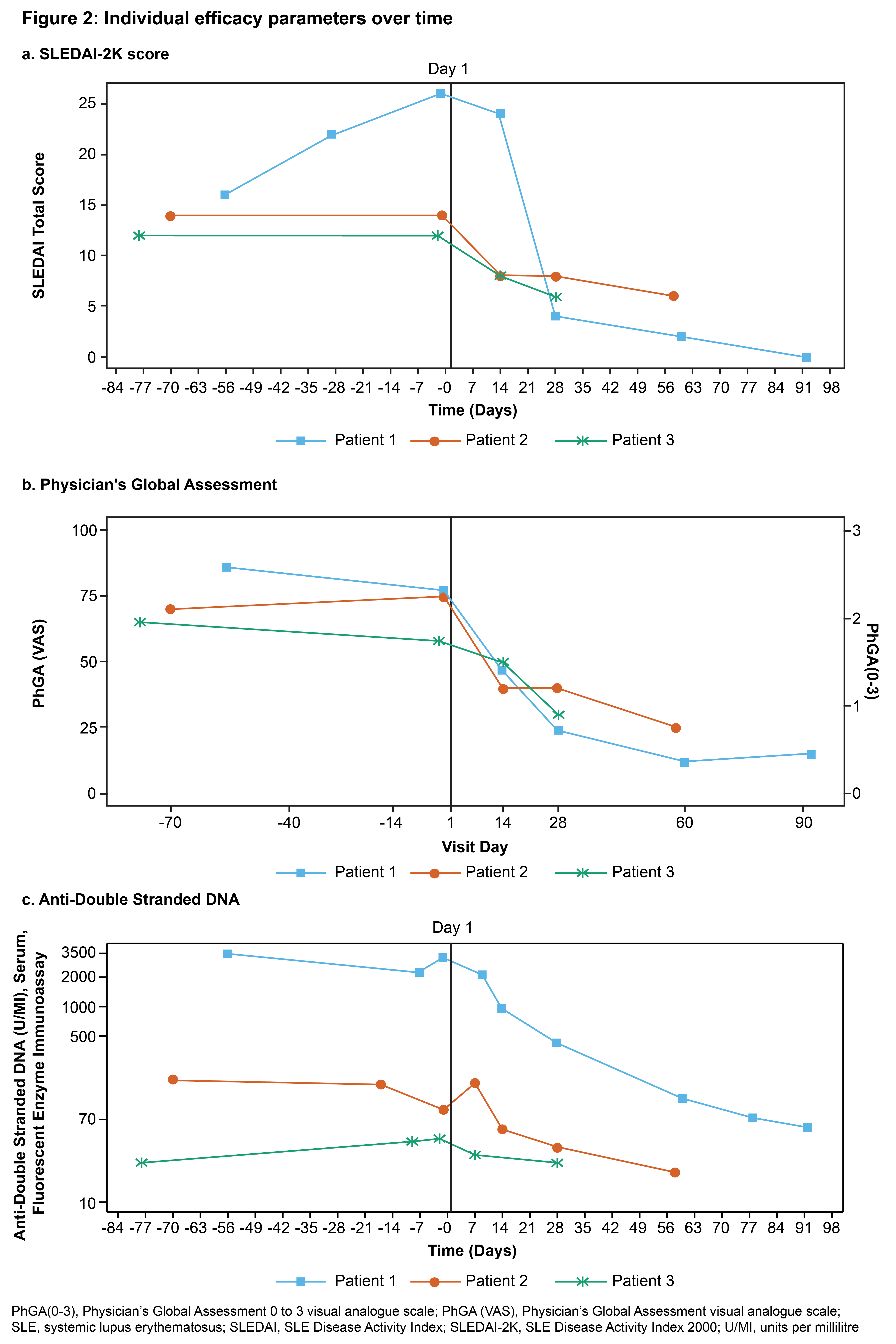
截图源于参考资料2
参考资料
1.https://ir.fatetherapeutics.com/news-releases/news-release-details/fate-therapeutics-presents-new-phase-1-clinical-data-ft819-shelf
2.https://acrabstracts.org/abstract/a-phase-1-multicenter-open-label-study-to-establish-the-preliminary-tolerability-efficacy-pharmacokinetics-and-pharmacodynamics-of-cc-97540-bms-986353-a-cd19-directed-car-t-cell-therapy-manufa/
3.https://acrabstracts.org/abstract/an-open-label-multicenter-phase-1-2-study-to-assess-safety-efficacy-and-cellular-kinetics-of-ytb323-a-rapid-manufacturing-car-t-cell-therapy-targeting-cd19-on-b-cells-for-severe-refractory-system/
4.https://www.cabalettabio.com/news-media/press-releases/detail/119/cabaletta-bio-presents-positive-clinical-safety-and
5.https://d1io3yog0oux5.cloudfront.net/_432762a6d6473a67982aabb26a44822b/cabalettabio/db/947/8249/pdf/Cabaletta+ACR+Poster+Presentation_20241108_final.pdf

相关推荐
随便看看
- CAR-T细胞产品SYS6020获批临床用于治疗系统性红斑狼疮
- CAR-T产品普基仑赛注射液纳入优先审评用于治疗CD19阳性的复发或难治性B细胞急性淋巴细胞白血病
- 超90%的患者肿瘤缩小!CAR-T疗法不仅治血癌,卵巢癌也有效!晚期患者也能实现肿瘤消失
- 中位生存20个月!CAR-T疗法横扫神经母细胞瘤、前列腺癌、胶质母细胞瘤
- CAR-T细胞疗法重击结直肠癌、甲状腺癌、卵巢癌等实体瘤
- 印度CAR-T细胞产品 talicabtagene autoleucel(Tali-cel™) I/Ib 期研究结果公布
- “抗癌神药”CAR-T疗法到底是班门弄斧还是真有奇效?免疫管家与你揭开谜底。
- 国内第六款CAR-T产品西达基奥仑赛获批上市!多发性骨髓瘤患者福音
- CAR-T细胞治疗:肿瘤免疫疗法的里程碑
- 伊基奥仑赛注射液治疗高危新诊断多发性骨髓瘤患者最新数据亮相2025 ASH











