
不知不觉中,免疫疗法已经占据了癌症治疗版图的“半壁江山”。从免疫检查点抑制剂到CAR-T细胞疗法,人类对抗癌症的武器正在不断进步。随着CAR-T细胞疗法在血液肿瘤领域“高歌猛进”时,科学家们将目光投向了更具挑战性的实体肿瘤战场。
实体瘤占癌症病例90%,始终是难以攻克的堡垒。T细胞受体工程化T细胞(TCR-T)疗法是癌症治疗的一种非常有前途的治疗方式,通过基因工程技术,将能够识别特定癌症抗原的TCR基因导入患者的T细胞中,使这些T细胞发挥特异性识别和杀伤肿瘤细胞的作用,以此来达到抗癌的目的。
目前国内TCR-T细胞临床试验正在寻找患者,患者可通过参加临床试验来接受药物治疗,年龄、疾病类型和阶段、药物史情况等符合临床试验“入选标准”的患者方可免费入组接受治疗。如果有需要或有兴趣了解临床试验的患者可以咨询康和源免疫之家医学部(400-880-3716)。
软组织肉瘤(STS)是来源于全身各部位结缔组织除骨或软骨以外的恶性肿瘤。其发病率相对较低,仅仅占恶性肿瘤的1%左右,常见的软组织肉瘤类型包括脂肪肉瘤、平滑肌肉瘤、横纹肌肉瘤、滑膜肉瘤、未分化多形性肉瘤和纤维肉瘤等。
TAEST16001是香雪制药研发的一款靶向NY-ESO-1的TCR-T细胞产品,有可能成为中国第一款上市的TCR-T细胞治疗药物。近日,一项关于TAEST16001治疗晚期软组织肉瘤的确证性临床试验方案专家研讨会召开,在该会议上,介绍了TAEST16001的I期临床试验数据。该试验涉及12名接受TAEST16001回输的晚期软组织肉瘤患者,这12名患者中41.7%的患者肿瘤缩小或消失。
而TAEST16001探索性II期临床试验中接受TAEST16001回输的8名晚期软组织肉瘤患者中,62.5%的患者肿瘤缩小或消失,客观缓解率(ORR)相较于I期试验数据有所提高。
TAEST16001的显著临床意义为软组织肉瘤患者提供了新的治疗选择,让软组织肉瘤患者临床缓解,减少病痛折磨的梦境得以实现。
这位脂肪肉瘤患者,在TCR-T细胞TAEST16001治疗前深受折磨,而TAEST16001治疗仅1个月,病情便保持稳定,2个月的时间便达到部分缓解(PR)状态,现已经持续缓解超1年!

截图源于Cell Reports Medicine
TBI-1301是宝生物株式会社自主研发的一款针对NY-ESO-1阳性肿瘤细胞TCR-T细胞疗法,2023年2月,《国家癌症杂志》发表了TBI-1301治疗滑膜肉瘤患者的1期临床数据。
3例患者中有2例患者在第12周达到病情稳定(SD)。其中患者1目标病灶先前接受过放射治疗,硬膜外病变缩小,肢体疼痛得到改善,该患者实现2年无进展生存。
患者3在第一次输注TBI-1301的前一天出现短暂进展,但幸运的是,TBI-1301输注后与前一天相比肿瘤缩小30%。
图片源于Int J Cancer
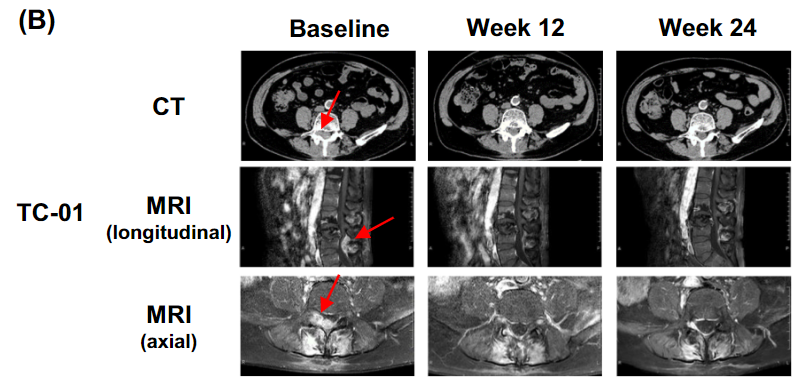
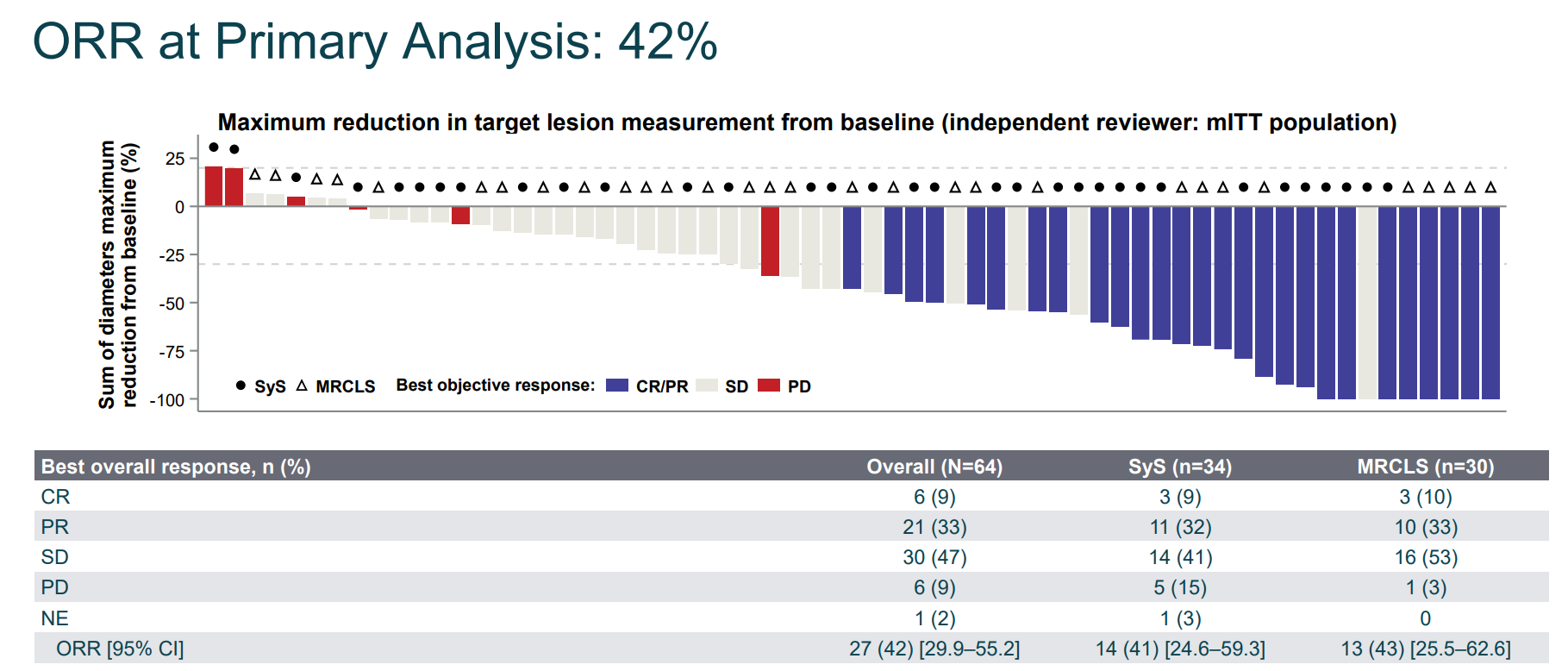
lete-cel是一种针对实体瘤抗原 NY-ESO-1 的TCR-T 细胞疗法,现已获得美国食品药品监督管理局(FDA)授予突破性疗法认定。在一项评估lete-cel在既往接受过治疗的晚期滑膜肉瘤或转移性粘液样/圆细胞脂肪肉瘤(MRCLS)患者中的疗效、安全性和耐受性的II 期 IGNYTE-ESO 试验中,评估了64名患者的疗效,结果显示:42%的患者获得临床反应,其中6例达到完全缓解(CR)状态,21例患者达到部分缓解状态,30例患者病情稳定(SD),中位缓解持续时间(DOR)为12.2个月。
34例滑膜肉瘤患者中有41%的患者获得临床反应,3例患者达到完全缓解(CR),11例患者部分缓解(PR),14例患者病情稳定(SD),中位缓解持续时间为18.3个月。
30例圆细胞脂肪肉瘤患者中有43%的患者获得临床反应,其中3例达到完全缓解,10例达到部分缓解,16例患者病情稳定,中位缓解持续时间为12.2个月。
肺癌是全球癌症相关死亡的主要原因。非小细胞肺癌是肺癌中最常见的类型,占85%以上。
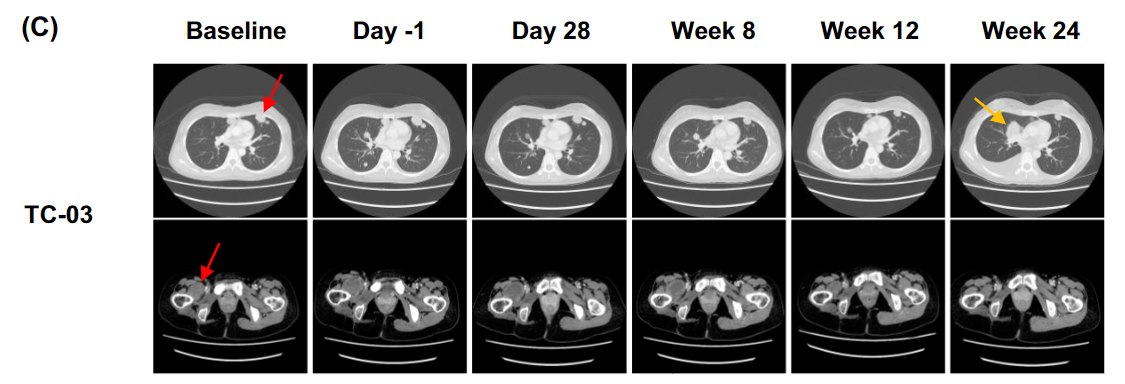
近期,纪念斯隆-凯特琳癌症中心研究人员在《CLINICAL CANCER RESEARCH》发表了lete-cel在晚期非小细胞肺癌中的两项初步研究数据。
研究 208749 是一项单臂研究,评估 lete-cel 在 IIIB 期或 IV 期或复发性 非小细胞肺癌患者中的安全性和临床活性。
研究208471是一项多臂Ib/IIa 期研究,评估 lete-cel 单独使用(A 组)或与 pembrolizumab(B 组和 C组)联合治疗 IIIB 期或 IV 期非小细胞肺癌患者的安全性和临床活性。
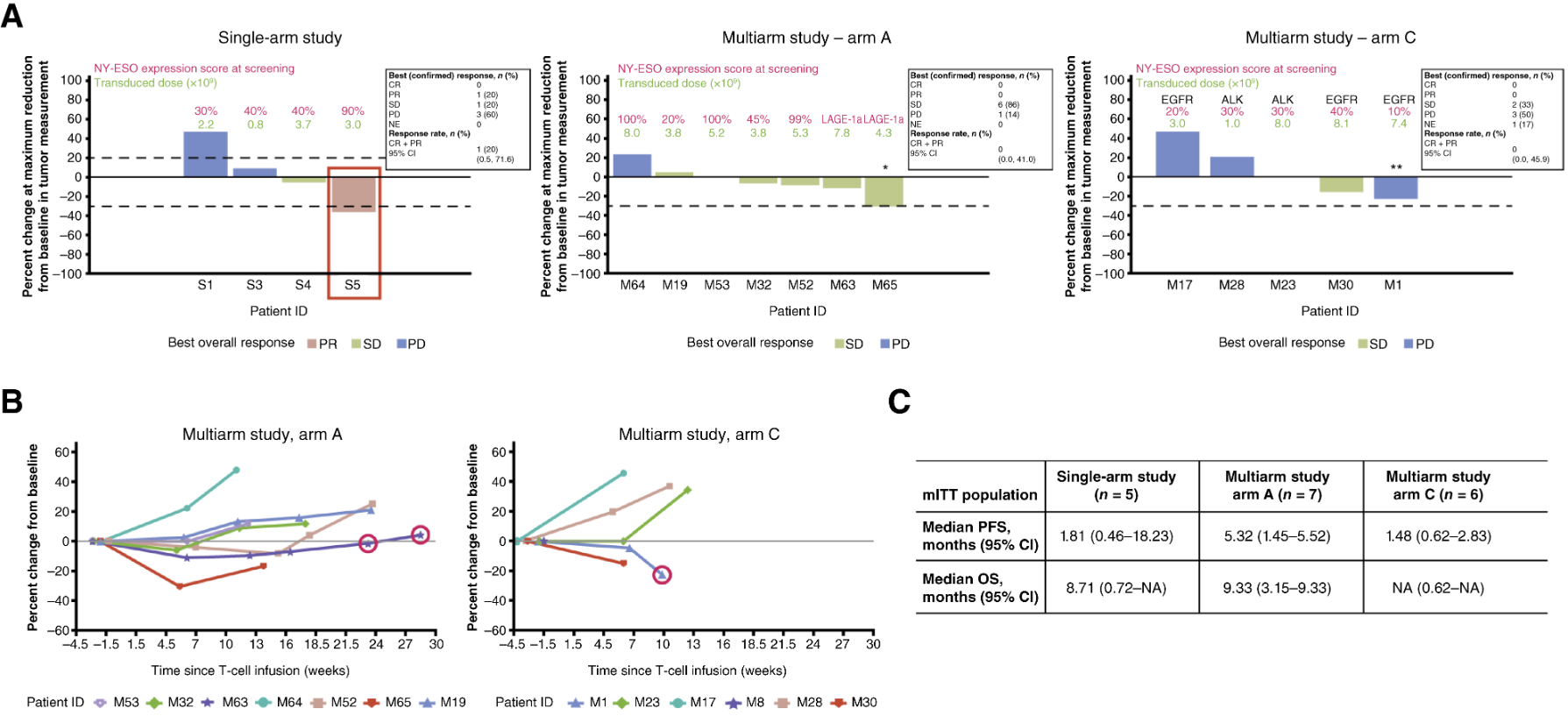
HLA-A*02 和抗原表达阳性的患者中,有 41 名患者进行单臂研究,9 例患者接受了白细胞去除术,其中 5 例患者接受了淋巴细胞清除和 T 细胞输注。5 例患者中有 1 例在首次 T 细胞输注后确认了部分缓解(PR),1例患者病情稳定(SD)。多臂研究中,共有 34 名患者接受了白细胞去除术,其中 13 名接受了淋巴细胞清除和 lete-cel 输注。lete-cel 单药治疗组中有6例患者病情稳定(SD),lete-cel 联合pembrolizumab组中有2例患者最佳反应为病情稳定,无患者出现部分缓解或完全反应。
图片源于Clin Cancer Res
单臂研究中患者S5既往接受过三线化疗和免疫疗法(nivolumab)治疗,第一次输注时该患者达到部分缓解(PR)状态,第一次输注后约28周,该患者接受第二次输注,达到病情稳定,非靶病变完全缓解。
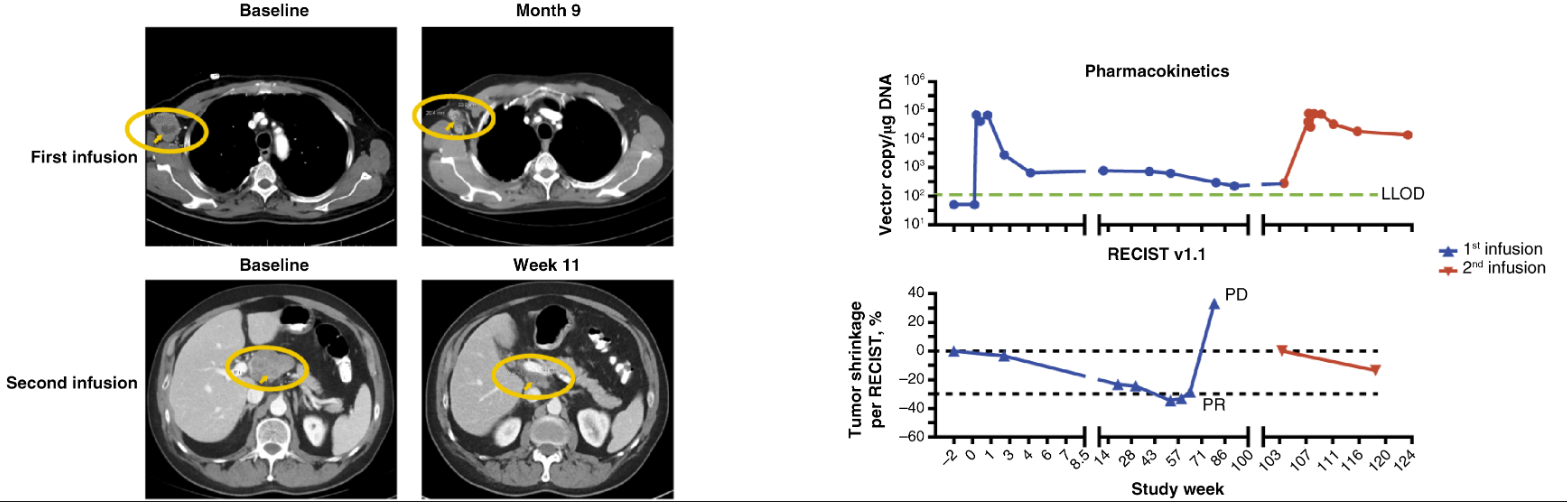
图片源于Clin Cancer Res
2018年10月,广东省深圳市第二人民医院研究人员在国际一流期刊《肿瘤学快报》(《Oncology Letters》)在线发表了TCR-T细胞治疗转移性非小细胞肺癌的病例报告。
2015年该患者在吉非替尼治疗2.5年后病情复发,伴有胸膜和肝转移。改用厄洛替尼治疗8个月后发现右侧肺门、纵隔、右侧胸膜、右侧肝叶和肝包膜疾病进展,于是参加了TCR-T细胞的临床试验 (NCT02457650)。幸运的是,T 细胞输注后第 43 天原发性肺肿瘤和肝转移便消退,原发性肺部病灶大小从 95×86×54 mm 减少到 64×44×54 mm,转移性肝脏病灶也从19.8×19.6×20 mm 减少到 10×10×10 mm。
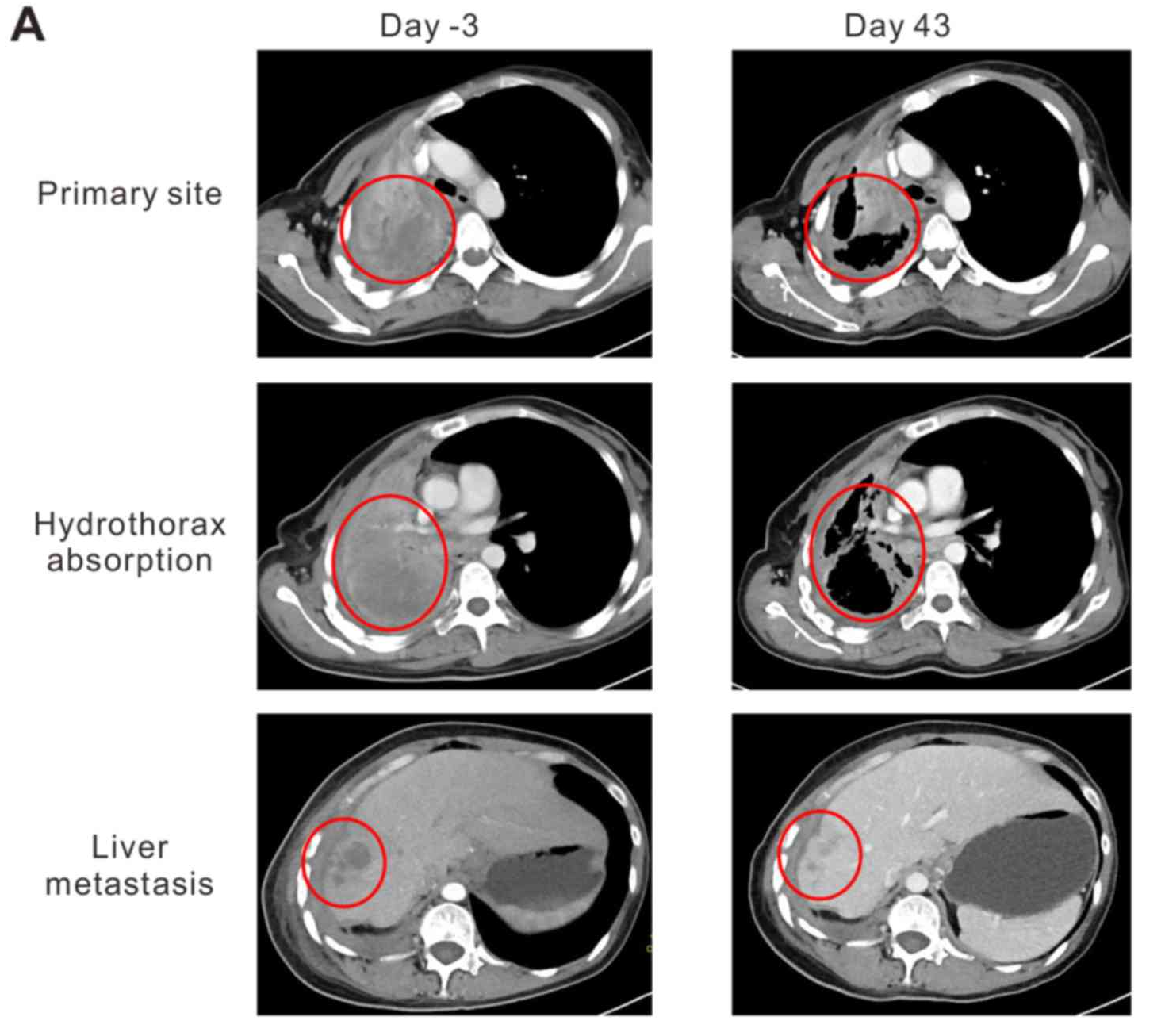
图片源于Oncology Letters
在人类癌症最常见的致癌基因中,突变型RAS影响约19%的肿瘤。携带突变型RAS的肿瘤细胞更具侵袭性,因此,与无RAS突变的患者相比,突变型RAS的肿瘤患者预后更差,总生存期(OS)较短。其中KRAS是最常见的突变亚型,在非小细胞肺癌、结直肠癌、胰腺导管腺癌、低级别浆液性卵巢癌(LGSOC)、子宫内膜癌中占主导地位。
胰腺癌是消化系统恶性程度最高的肿瘤之一,5年生存率约10%,被称为“万癌之王”。近几年,胰腺癌发病率呈现上升趋势,在所有恶性肿瘤中,它的发病率排在前十名。
2022年,医学期刊《新英格兰医学杂志》(NEJM)发布了美国Eric Tran团队通过TCR-T细胞疗法成功治疗KRAS G12D突变的胰腺癌的案例。
该患者确诊为胰头腺癌,接受了4个周期的FOLFIRINOX的新辅助化疗方案并进行了手术切除治疗,然后再次化疗。这次治疗让她有一段时间没再患病,但是2019年,她出现了肺转移。许多癌症患者在传统治疗方式均束手无策后,细胞免疫治疗便成为他们最后一根救命稻草。为此,该患者于2021年6月接受了自体外周血T细胞治疗。输注1个月后,患者肺部转移病灶大幅度消退,肿瘤缩小62%。细胞输注仅仅半年的时间,肿瘤已经缩小了72%。
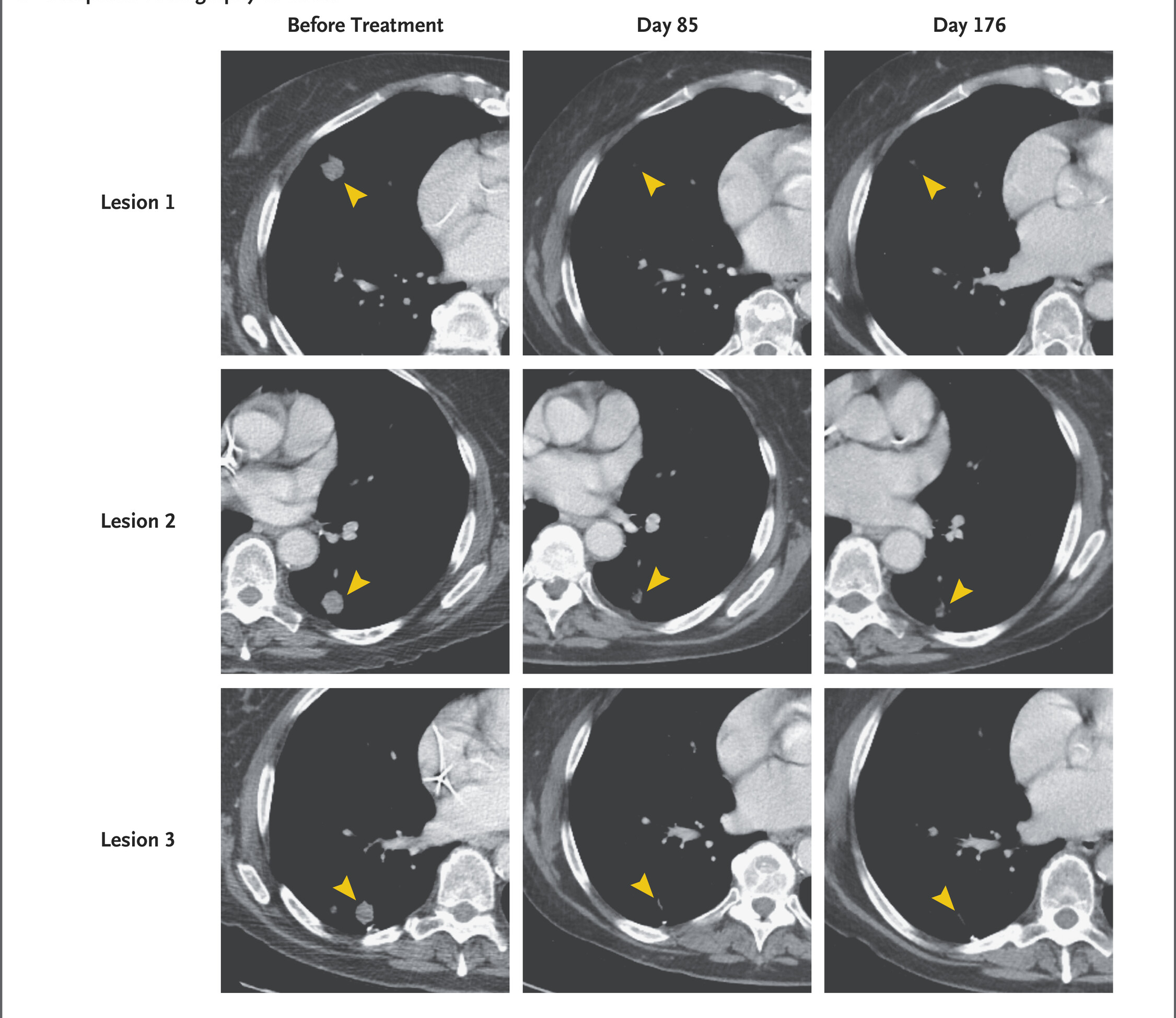
图片源于NEJM
目前康和源免疫之家有靶向KRAS突变的TCR-T细胞疗法的临床试验正在寻找肠癌患者,患者可通过参加临床试验来接受药物治疗,病历资料审核通过后可免费入组接受治疗。如果有需要或有兴趣了解临床试验的患者可以咨询康和源免疫之家医学部(400-880-3716)。 部分入选标准: 1.年龄18岁至70周岁; 2.至少有一个可测量病灶; 3.ECOG评分0-1分。 临床试验对于传统疗法无效或疗效较差的患者来说,可能带来新的希望和治疗机会,并能大大减轻家庭经济负担。对临床试验有兴趣或需要帮助的患者可提交病历资料至康和源免疫之家医学部(400-880-3716)来寻找适合的临床研究。
目前TCR-T细胞治疗的临床试验覆盖恶性黑素色瘤、尿路上皮癌、直肠癌、食管癌、滑膜肉瘤等实体瘤,并在美国上市一款用于治疗滑膜肉瘤的细胞治疗产品。国内企业也正在积极地跟进国际步伐,香雪制药的TAEST16001在肉瘤患者中已经表现出令人鼓舞的治疗效果,有望成为中国第一款上市的TCR-T细胞治疗药物。随着科学技术的不断进步和临床试验的深入,TCR-T细胞疗法有可能成为治疗癌症的重要手段,期待更多如TCR-T、CAR-T的疗法获批上市,让患者有更加精准、多样的治疗选择! 参考资料 1.Pan, Qiuzhong et al.”Phase 1 clinical trial to assess safety and efficacy of NY-ESO-1-specific TCR T cells in HLA-A∗02:01 patients with advanced soft tissue sarcoma’Cell Reports Medicine, Volume 4, Issue 8, 101133 https://www.cell.com/cell-reports-medicine/fulltext/S2666-3791(23)00261-6 2.Ishihara M, Nishida Y, Kitano S, et al. A phase 1 trial of NY-ESO-1-specific TCR-engineered T-cell therapy combined with a lymph node-targeting nanoparticulate peptide vaccine for the treatment of advanced soft tissue sarcoma. Int J Cancer. 2023; 152(12): 2554-2566. doi:10.1002/ijc.34453 3.https://d1io3yog0oux5.cloudfront.net/_d32e7779516fa58c59f9d4e9d7ddb9a0/adaptimmune/files/documents/DAngelo_IGNYTE-ESO_CTOS24.pdf 4.Mehmet Altan, Gilberto Lopes, T. Jeroen N. Hiltermann, Ramaswamy Govindan; Safety and Tolerability of Letetresgene Autoleucel (GSK3377794): Pilot Studies in Patients with Advanced Non–Small Cell Lung Cancer. Clin Cancer Res 1 February 2025; 31 (3): 529–542. https://doi.org/10.1158/1078-0432.CCR-24-1591 5.Xia, Y., Tian, X., Wang, J., Qiao, D., Liu, X., Xiao, L., Liang, W., Ban, D., Chu, J., Yu, J., Wang, R., Tian, G., Wang, M."Treatment of metastatic nonsmall cell lung cancer with NYESO1 specific TCR engineeredT cells in a phase I clinical trial: A case report". Oncology Letters 16, no. 6 (2018): 6998-7007. https://doi.org/10.3892/ol.2018.9534 6.Rom Leidner, Nelson Sanjuan Silva, Huayu Huang, et al“Neoantigen T-Cell Receptor Gene Therapy in Pancreatic Cancer”.N Engl J Med 2022;386:2112-2119
扫描下方二维码,或者直接电话咨询康和源免疫之家医学部(400-880-3716),持续为您关注并分享最新消息。

免责声明:文本参考来源于网络,版权归原作者所有。
该文章仅供分享,如涉嫌侵犯您的著作权请联系我们删除,谢谢!
相关推荐
随便看看
- FDA加速批准!TCR-T疗法让胰腺癌患者半年时间肿瘤缩小72%,94%患者病毒暴跌
- 肿瘤缩小超60%!美国FDA认证官方的TCR-T疗法为实体瘤患者迎来新希望,8例患者7例显效
- 无瘤生存超9年!TCR-T细胞疗法效果更上一层楼!
- TCR细胞疗法与HLA
- TCR-T细胞治疗实现宫颈癌患者完全缓解!SCG142表现出优异的抗肿瘤活性
- 肿瘤靶病灶缩小74.5%!我国TCR-T细胞疗法蓬勃发展,剑指肝细胞癌、肉瘤、胰腺癌、结直肠癌等
- 70%的患者治疗两年后仍然存活!全球首款TCR-T细胞疗法获FDA优先审评资格,滑膜肉瘤患者新的治疗选择
- 疾病控制率达到64%,TCR-T疗法有望成为实体瘤中的“王牌战队”
- 镔铁生物TCR-T细胞治疗产品IX001 TCR-T注射液获得临床试验许可,用于胰腺癌
- IX001 TCR-T注射液实现首例入组胰腺癌患者部分缓解











