
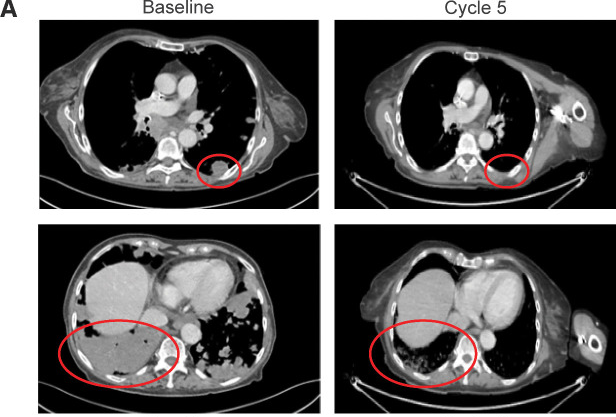
截图来源于“参考资料2” 参考资料 1.Bristol Myers Squibb - U.S. Food and Drug Administration Approves Augtyro™ (repotrectinib), a Next-Generation Tyrosine Kinase Inhibitor (TKI), for the Treatment of Patients with NTRK-Positive Locally Advanced or Metastatic Solid Tumors (bms.com) 2.FDA grants accelerated approval to repotrectinib for adult and pediatric patients with NTRK gene fusion-positive solid tumors | FDA 3.Molecular Characteristics of Repotrectinib That Enable Potent Inhibition of TRK Fusion Proteins and Resistant Mutations - PMC (nih.gov) 4.Efficacy and Safety of Larotrectinib in Patients With Tropomyosin Receptor Kinase Fusion–Positive Lung Cancers - PMC (nih.gov) 5.Updated efficacy and safety of entrectinib in NTRK fusion-positive non-small cell lung cancer - Lung Cancer (lungcancerjournal.info)





相关推荐
随便看看
- 绿叶制药小细胞肺癌创新药赞必佳®(注射用芦比替定)获批上市
- 美国FDA批准cosibelimab用于皮肤鳞状细胞癌
- 卡度尼利单抗获批宫颈癌的新适应症
- CDK4/6抑制剂泰瑞西利胶囊III期临床研究成果亮相《JAMA Oncology》
- ADC药物TIVDAK、DS-8201新突破!满足宫颈癌、胃癌患者需求
- IBI363获快速通道资格拟用于治疗黑色素瘤
- 2025年有望在国内获批上市的ADC药物,重击乳腺癌、肺癌、鼻咽癌等
- PLN-101095 在免疫检查点抑制剂难治性晚期实体瘤患者中的中期 1 期数据公布
- 新一代ADC药物(ADRX-0405)获美国FDA孤儿药资格认定
- ADC药物JSKN003获得美国FDA授予快速通道资格认定











